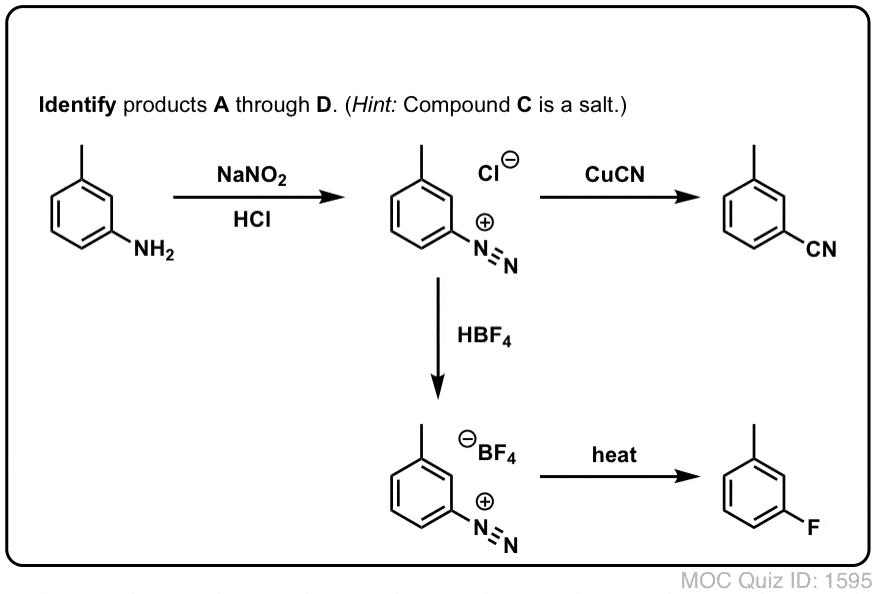
![Compound [A] is an aromatic amine which react with NaNO2 + HCl at 273 - 278 K and form compound [B] . Compound [B] react with HBF4 and the obtain product on Compound [A] is an aromatic amine which react with NaNO2 + HCl at 273 - 278 K and form compound [B] . Compound [B] react with HBF4 and the obtain product on](https://haygot.s3.amazonaws.com/questions/774137_738479_ans_d47545d3f5ad465d9fd600a977e7df56.png)
Compound [A] is an aromatic amine which react with NaNO2 + HCl at 273 - 278 K and form compound [B] . Compound [B] react with HBF4 and the obtain product on

Organic Solid Acid/NaNO2: An Efficient System for the Oxidation of Urazoles and Bis-Urazoles under Mild and Heterogeneous Conditions

NaNO2–Ceric ammonium nitrate mediated conversion of acrylic esters and Baylis–Hillman derived acrylic esters into corresponding β-nitro acrylic esters - ScienceDirect
Scheme 1: Reagents and conditions: (a) NaNO2/HCl,-5˚C, 1 hr; (b) ethyl... | Download Scientific Diagram

NaNO2/I2 as an alternative reagent for the synthesis of 1,2,3-benzotriazin-4(3H)-ones from 2-aminobenzamides - ScienceDirect

Le nitrite de sodium de qualité industrielle Nano2 Le fabricant - Chine Le nitrite de sodium, le nitrite de sodium Tech Grade

NaNO2/I2‐Mediated Regioselective Synthesis of Nitrosoimidazoheterocycles from Acetophenones by a Domino Process - Mukhopadhyay - 2016 - European Journal of Organic Chemistry - Wiley Online Library

Scheme 1. Reagents and conditions: (a) (i) NaNO2, HCl, 0 °C, 30 min;... | Download Scientific Diagram

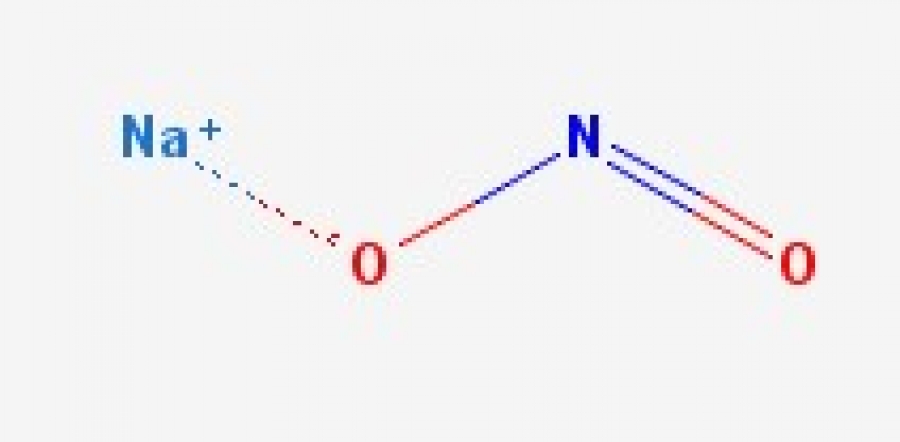
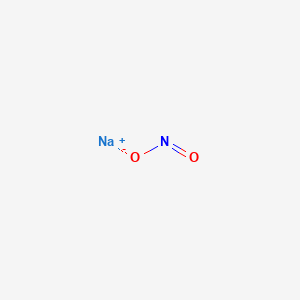
Nitrite de sodium, molécule de NaNO2. Il est utilisé comme conservateur alimentaire et antidote à l'empoisonnement au cyanure. Formule chimique structurelle et modèle de molécule. V Image Vectorielle Stock - Alamy
![Sodium Nitrite [NaNO2] 99.6% ACS Grade Powder 1 Lb in Two Space-Saver Bottles USA : Amazon.in: Industrial & Scientific Sodium Nitrite [NaNO2] 99.6% ACS Grade Powder 1 Lb in Two Space-Saver Bottles USA : Amazon.in: Industrial & Scientific](https://m.media-amazon.com/images/I/91z1BiNBE4L._SL1500_.jpg)
Sodium Nitrite [NaNO2] 99.6% ACS Grade Powder 1 Lb in Two Space-Saver Bottles USA : Amazon.in: Industrial & Scientific